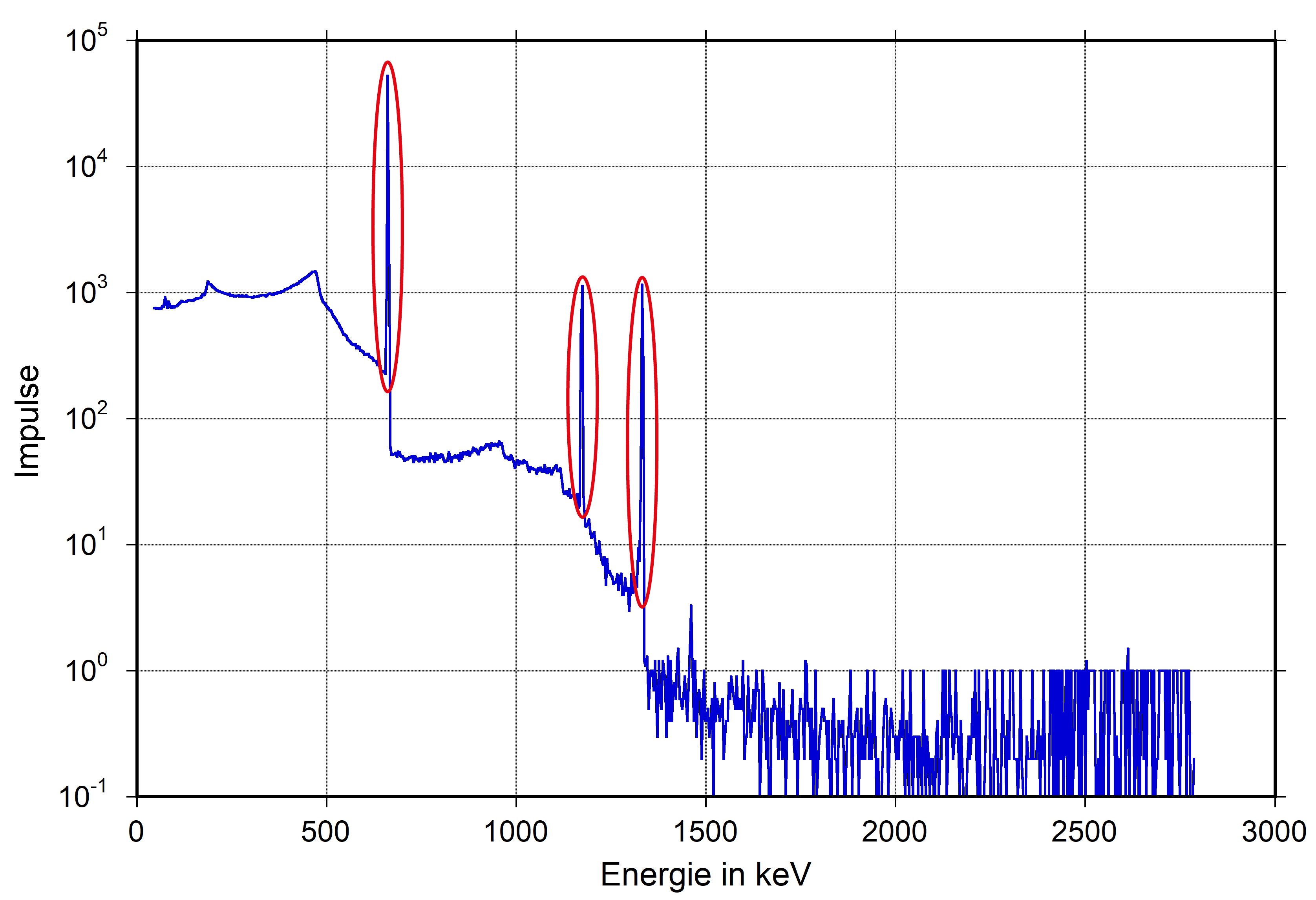
Gamma Spectra
These increases in the diagrams are referred to as Peaks.
By the way:
These diagrams are called spectrum. We will use this term in the following sections as well.
a diagram = a spectrum
multiple diagrams = multiple spectra
The special thing about gamma radiation is that almost every radioactive nuclide emits radiation with one or more distinct energies. This allows for identification (clearly) based on the measured peaks.
These peaks are referred to as characteristic lines. They can be compared to a fingerprint, which is different for any two individuals. In this comparison, the individuals are the radionuclides, and the fingerprints are the characteristic lines of a radionuclide.
The evaluation of a spectrum, i.e., the identification (determining which radionuclides are present in a spectrum) is somewhat similar to detective work.
Now it’s your turn! On the next page, we will show you measured spectra for various radioactive nuclides. The captions indicate the respective nuclide as well as the energy of the characteristic line(s) of that nuclide. Try to find the peaks in the respective spectrum using this information - you should not find it too difficult if you have read the content of the previous pages and answered the corresponding questions correctly. If you are unsure, then read through the section What information does a gamma measurement provide again.
 At the end of the next page, you will be shown a spectrum from a measurement of a sample - it could, for example, be the container from the introductory chapter - without any indication of the radionuclides. Your task will be to identify the nuclides contained in the container based on the characteristic lines. With the knowledge you have gathered up to this task, you should find that easy.
At the end of the next page, you will be shown a spectrum from a measurement of a sample - it could, for example, be the container from the introductory chapter - without any indication of the radionuclides. Your task will be to identify the nuclides contained in the container based on the characteristic lines. With the knowledge you have gathered up to this task, you should find that easy.


